New Applications

Submit A New Application
To obtain a Certificate of Suitability to the monographs of the European Pharmacopoeia (CEP), applicants must send in electronic format the following documentation to the Certification of Substances Department (DCEP) of the EDQM ...

Content of the Dossier
Detailed information on what an application should contain is described in the documents below. Refer to the document relevant to your application.
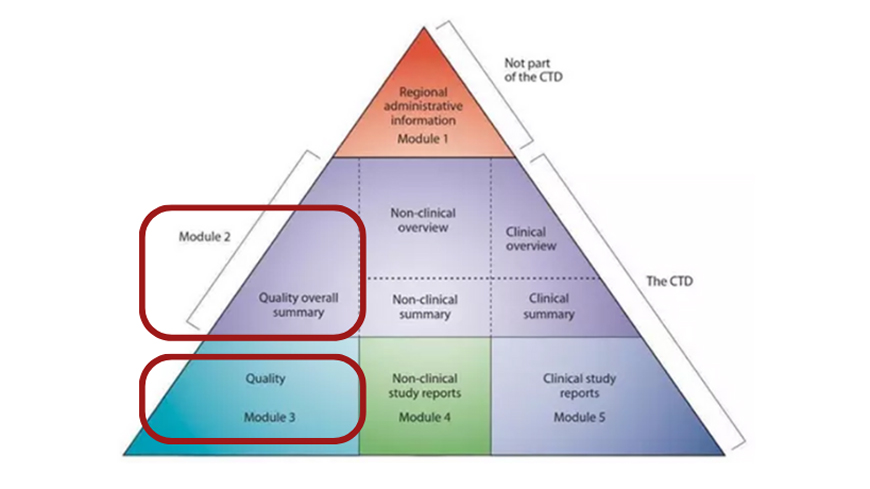
Quality Overall Summary (QOS) for CEP applications
The Quality Overall Summary (QOS) plays a crucial role in the Certificate of Suitability to the European Pharmacopoeia monographs (CEP) procedure.

Top Ten Deficiencies in New Applications for Certificates of Suitability for Chemical Purity
A summary of the top ten deficiencies identified after the initial evaluation of new applications for Certificates of Suitability (CEP) for chemical purity.

Sister files
The sister file procedure is intended to facilitate the submission of similar dossiers within the Certification Procedure, and to allow applicants to benefit from a fast-track procedure and harmonised assessments ...

Submission format & e-submission
Applicants should send their documentation in electronic format only to the Certification of Substances Department (DCEP) of the EDQM.
- Regulatory Reliance and Fast track assessment in the CEP procedure
- Tips to improve the quality of your dossier and get a CEP faster
- Top Ten Deficiencies in New Applications for Certificates of Suitability for Chemical Purity PA/PH/CEP (24) 10
- New requirements for the content of the CEP dossier for chemical purity and for herbal drugs/herbal drug preparations according to the CEP 2.0
- How CEP holders can avoid the rejection of notifications
- Change in contact details for a certificate of suitability application (to be used from 1 April 2022)
- CEP holders responsibilities towards their customers (PA/PH/CEP (21) 57, January 2022)
Please go to Certification Policy documents & Guidelines for a list of all of our guidelines.
Search for a list of granted CEPs, their type, the name of the substance, the full CEP number, the issue date and validity status, using the Certification Database (updated daily).
Applicants commit to provide samples of substance and/or impurities when requested by the EDQM.




